Meet Morganella morganii: A Familiar Stranger in Your Gut
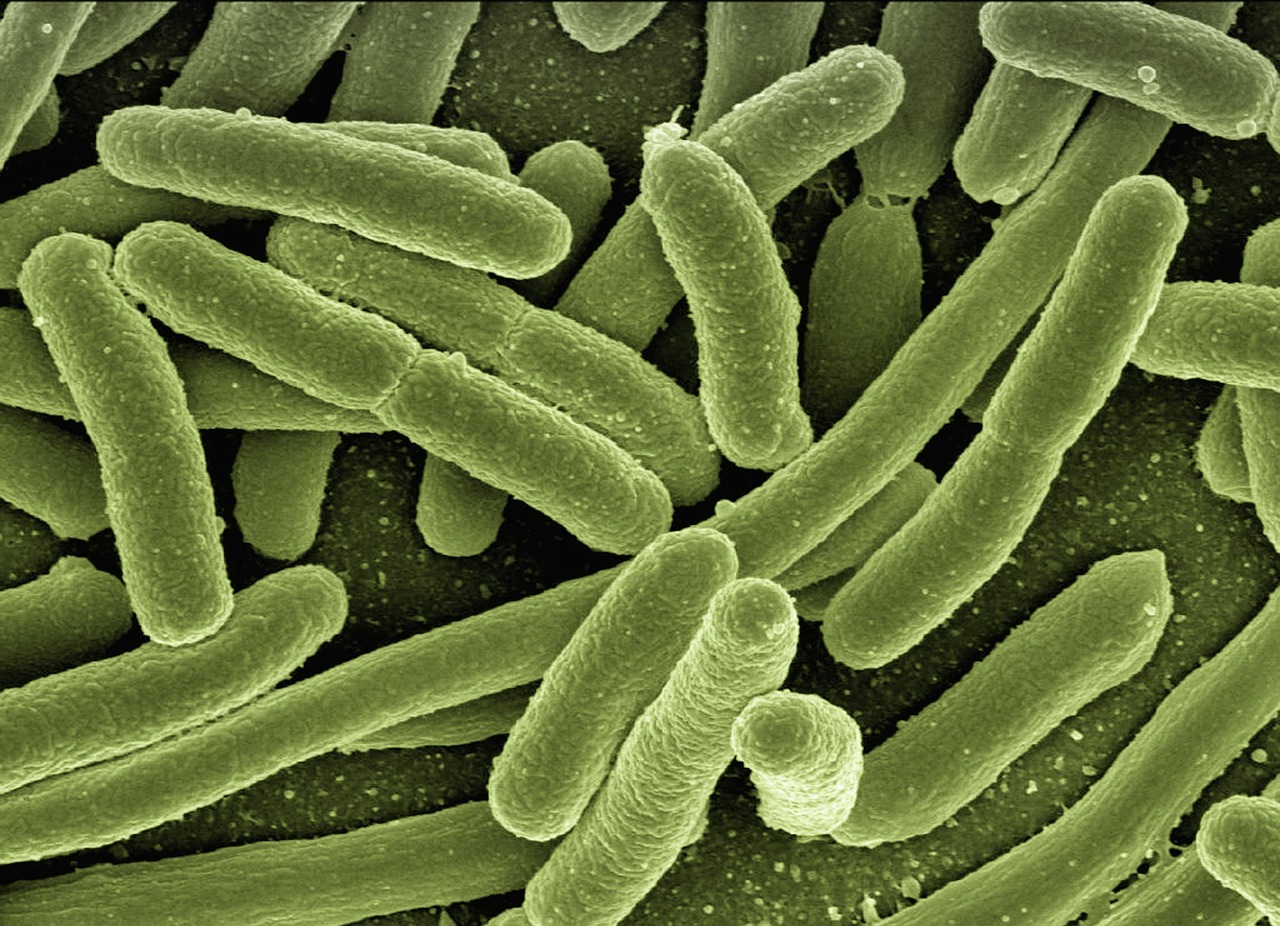
Morganella morganii is a species of Gram-negative bacteria that has a commensal relationship within the intestinal tracts of humans, mammals, and reptiles as normal flora. Although it has a wide distribution, it is considered an uncommon cause of community-acquired infection, and is most often encountered in postoperative and other nosocomial infections. In other words, it’s been quietly living in human guts for a very long time, mostly going unnoticed.
A particularly robust and intriguing study that employed genetic, diet, disease, and microbiota data from several thousand individuals identified a strong association between Morganella morganii and major depressive disorder. While the strongest association of M. morganii is with MDD, its abundance is also associated with inflammatory diseases, including type 2 diabetes. M. morganii, a Gram-negative facultative anaerobe found in both the environment and intestinal tracts, is conventionally regarded as an opportunistic pathogen, and recent studies have reported its ability to produce DNA-damaging metabolites linked to inflammatory bowel disease.
One bacterium in particular, Morganella morganii, has been linked in several studies to major depressive disorder. Until recently, though, it was unclear whether this microbe contributes to depression, whether depression changes the microbiome, or whether another factor explains the connection. That ambiguity is precisely what the new Harvard research set out to resolve.
The Harvard Study That Changed the Conversation

A team from Harvard Medical School has now identified a biological mechanism that strengthens the case for M. morganii’s role in brain health. Their findings offer a plausible explanation for how this bacterium may influence mental health. Published on January 16 in the Journal of the American Chemical Society, the study points to an inflammation-triggering molecule that could serve as a potential biomarker for diagnosing or treating some cases of depression.
Sunghee Bang and Yern-Hyerk Shin led this study at Harvard Medical School and the Broad Institute, collaborating with Jon Clardy’s group. The study is published in the Journal of the American Chemical Society. The collaboration brought together specialists in small-molecule chemistry and microbiome immunology, which turned out to be exactly the right combination for this kind of discovery.
This work was funded by the National Institutes of Health (grant R01AI172147) and The Leona M. and Harry B. Helmsley Charitable Trust. That’s a notable level of institutional backing for research exploring a connection that, even a few years ago, most psychiatrists would have found surprising at best.
The Role of an Industrial Pollutant: Diethanolamine

The researchers discovered that an environmental contaminant called diethanolamine, or DEA, can sometimes replace a sugar alcohol in a molecule produced by M. morganii in the gut. This altered molecule behaves very differently from the normal version. Instead of remaining harmless, it activates the immune system, prompting the release of inflammatory proteins known as cytokines, especially interleukin-6 (IL-6).
The role of diethanolamine in this study highlights the broader impact of environmental micropollutants on human biology. DEA is widely used in industrial processes and personal care products, and its presence in the environment means exposure is nearly unavoidable for most people in developed countries. When a common bacterium like M. morganii transforms DEA into an immune-stimulating molecule, it creates a new layer of interaction between pollution, the microbiome, and the immune system.
DEA is not known to be a natural part of either human or bacterial biochemistry. Instead, it is an industrial contaminant whose ability to be incorporated at low levels into animal and human lipids through the apparently not-so-picky enzyme cardiolipin synthase has been confirmed for decades. That the body has been quietly absorbing this chemical all along, without anyone connecting it to mental illness, is one of the more striking aspects of this story.
The Unusual Phospholipids at the Heart of the Discovery

A bioassay-guided fractionation approach identified bacterially produced metabolites that induced pro-inflammatory immune responses. The metabolites are unusual phospholipids that resemble conventional cardiolipins, in which diethanolamine replaces the central glycerol. These molecular chimeras of endogenous metabolites from phospholipid biosynthetic pathways and the industrially produced micropollutant DEA activate TLR2/TLR1 receptors and induce the production of pro-inflammatory cytokines, especially IL-6.
That fatty molecule belongs to a group called cardiolipins, which are known to stimulate cytokine release. In the new study, researchers found that when DEA is incorporated into the molecule produced by M. morganii, it begins to behave like a cardiolipin, triggering inflammation. Cardiolipins are molecules the immune system is primed to respond to, so this chemical impersonation has real consequences inside the body.
Only the versions of these lipids that contained DEA caused a strong IL-6 response. It turns out that small changes in the shape of the fatty acid chains, like having a double bond instead of a ring, made a big difference in how strongly the immune system reacted. This shows that the immune system can pick up on very fine molecular details in bacterial chemicals.
How the Immune System Becomes a Bridge to the Brain
When M. morganii adds a chemical called DEA to its fats, it changes the molecule in a way that triggers the body’s immune system. Tests showed that this new molecule turns on special sensors in immune cells called TLR1 and TLR2. Once these sensors are activated, the immune cells release a substance called interleukin-6 (IL-6).
Clardy and colleagues found that conversion of a common environmental contaminant known as diethanolamine, or DEA, by a species of gut bacteria, Morganella morganii, stimulates the release of cytokines, particularly interleukin-6 (IL-6). Not only has M. morganii been linked to inflammatory conditions such as type 2 diabetes and inflammatory bowel disease, but IL-6 has specifically been connected to major depressive disorder.
Chronic inflammation is known to play a role in many diseases and has also been associated with major depressive disorder. The IL-6 link matters because this cytokine doesn’t stay put in the gut. It can reach the brain through multiple pathways, influencing mood, cognition, and behavior in ways researchers are still working to fully map.
What Made This Study’s Association Signal So Convincing
This earlier study identified M. morganii as having a possible causal role in major depressive disorder. It was one of the strongest causal signals in the team’s genome-wide association analysis involving 2,801 microbial taxa and 7,967,866 human genetic variants from 5,959 individuals, and it was the only one that validated when they went back and checked multi-year health records taken after the original microbiota samples. That kind of replication across time gives the association considerably more weight than a simple one-point correlation.
Correlational studies linking gut microbes and human phenotypes have frequently identified strong statistical associations but not their underlying molecular mechanisms. A particularly robust and intriguing study that employed genetic, diet, disease, and microbiota data from several thousand individuals identified a strong association between Morganella morganii and major depressive disorder.
Published in the Journal of the American Chemical Society, the study points to an inflammation-triggering molecule and suggests a possible new target for diagnosing or treating certain cases of depression. It also provides a framework for studying how other gut microbes may shape human health and behavior.
Depression as an Immune Condition: A Shifting Paradigm

The bacterium incorporates an environmental contaminant into one of its molecules, triggering inflammation, which is a known factor in disease development. These findings suggest the contaminant could serve as a biomarker and further support the idea that major depressive disorder may have autoimmune connections.
Accumulating studies have shown an association between dysbiosis and depression by reporting changes in the gut microbial composition of major depressive disorder patients compared to healthy individuals, particularly in terms of microbial diversity and the relative abundance of specific bacterial taxa. Enriched pro-inflammatory bacteria and depletion of anti-inflammatory bacteria have been the most consistent findings.
The findings add weight to the idea that depression, or at least some forms of it, may involve the immune system. That’s a genuine conceptual shift. For years, major depressive disorder has been treated almost exclusively as a neurological or neurochemical problem. The idea that the immune system, nudged by a gut microbe and an industrial pollutant, could be an equal player changes how doctors and researchers might frame treatment.
The Broader Gut-Brain Axis: Context for This Finding
Recent studies underscore the central role of gut microbiota in modulating neurological and psychological health via the gut-brain axis. Key findings highlight the importance of dietary components, including probiotics, prebiotics, and psychobiotics, in restoring microbial balance and enhancing mood regulation. Different dietary patterns exhibit a profound impact on gut microbiota composition, suggesting their potential as complementary strategies for mental health support. Furthermore, mechanisms like tryptophan metabolism, the HPA axis, and microbial metabolites such as short-chain fatty acids are implicated in linking diet and microbiota to depression.
A study investigating the relation of fecal microbiome diversity and composition with depressive symptoms in over a thousand participants from the Rotterdam Study cohort, and validated in over fifteen hundred subjects, identified associations with thirteen microbial taxa. These bacteria are known to be involved in the synthesis of glutamate, butyrate, serotonin, and gamma amino butyric acid, which are key neurotransmitters for depression. The Morganella finding slots into this larger picture, adding a specific, traceable mechanism where before there were only patterns.
Depression, a highly prevalent and relapsing mental disorder, exacts profound personal and socioeconomic tolls globally, warranting urgent scientific and clinical attention. Emerging evidence from both preclinical models and human clinical investigations has established the microbiota-gut-brain axis as a critical determinant in depression pathogenesis.
DEA as a Potential Biomarker: What That Could Mean for Diagnosis

The researchers suggest that DEA could potentially be used as a biomarker to help identify certain cases of major depressive disorder. This is a practical implication of the chemistry, not just academic curiosity. If DEA levels in the body correlate with higher M. morganii activity and higher IL-6 release, it might one day be possible to test for a subtype of depression using a blood or stool marker rather than relying entirely on symptom checklists.
Chronic inflammation is known to play a role in many diseases and is also associated with major depressive disorders. This is leading to a new pathway to treating depression, as DEA could potentially be used as a biomarker to help identify some cases of major depressive disorder. The value of that kind of precision can’t be overstated, given how many patients cycle through different antidepressants before finding something that works.
Researchers note that while the mechanism is clear, further work is needed to determine how often this occurs in depressed individuals and whether blocking this pathway alleviates symptoms. They plan to explore whether other gut microbes use similar chemical substitutions and what portion of depression cases might involve this contaminant-inflammation route.
What Comes Next: Treatment Possibilities on the Horizon

Experts believe these findings will inspire personalized treatment approaches tailored to an individual’s unique gut microbiome composition. Future interventions might include probiotics, dietary modifications, or drugs designed to inhibit harmful bacterial functions and inflammatory responses without solely focusing on altering brain chemistry. Although further research is necessary before these insights can be translated into clinical practice, this advancement marks a significant step toward more holistic depression treatment.
This finding helps explain how gut microbes can influence brain health at a molecular level. It also raises the possibility of new treatments that target the immune system rather than just the brain. That’s a meaningful redirection. Current antidepressants work on neurotransmitter systems and help a lot of people, but a meaningful share of patients don’t respond adequately to them. Immune-targeted approaches could potentially reach those who don’t.
The integration of synthetic biology, including CRISPR-modified psychobiotics, nanotechnology, and computational psychiatry is on the horizon, and could usher in a new era of microbiome-based depression therapies. Realizing the full potential of these approaches will demand interdisciplinary collaboration across fields like microbial genomics, neuroimmunology, and computational sciences. By bridging mechanistic insights with clinically actionable solutions, these innovations hold the potential to revolutionize the treatment of mental health disorders.
What This Discovery Does and Doesn’t Prove
More research will be needed to determine whether this altered molecule directly causes depression and to understand how many cases might be influenced by this process. That caveat matters. The Harvard team identified a credible, testable molecular mechanism, but they haven’t proven that every depressed person has elevated M. morganii, or that removing DEA from the gut would reliably lift someone’s mood.
Morganella morganii has been linked to major depressive disorder in several studies. Until now, it was unclear whether this bacterium contributes to the disorder, whether depression alters the microbiome, or if another factor is involved. The Harvard work significantly sharpens that picture, but it doesn’t close the case entirely. Science rarely does in a single paper.
By revealing how a gut bacterium turns a common contaminant into a pro-inflammatory agent, the study offers new insight into the complex makeup of mental health disorders, and hints at novel ways to detect and treat them. The honest reading of this research is that it opens a door, a well-constructed, biochemically rigorous door, rather than offering a final answer to one of medicine’s most stubborn questions.
Conclusion: A Gut Feeling That Turned Out to Be Science

This research tells a coherent story from M. morganii at the beginning to depression at the end, as the authors propose, since chronic inflammation contributes to the development of many diseases and has been linked with depression. The connection is further strengthened by previous studies associating IL-6 with major depressive disorder and linking M. morganii with inflammatory conditions such as type 2 diabetes and inflammatory bowel disease.
This breakthrough was made possible by combining expertise from two research groups. The Clardy Lab focuses on the chemistry of small molecules produced by bacteria, while the lab of Ramnik Xavier specializes in understanding how the microbiome affects health at a molecular level. Together, these collaborations have advanced the understanding of how gut bacteria interact with the immune system and influence disease.
Depression has resisted neat explanations for most of modern medicine’s history. What the Morganella research suggests is that the answer may have been, quite literally, living inside us all along. The stomach and the brain are not separate territories. They’re in constant conversation, and sometimes what happens in one shapes everything about the other. That’s not a poetic metaphor anymore. It’s biochemistry.


