Everything we learned in school about life painted a very specific picture. Oxygen in, carbon dioxide out. Breathe, metabolize, repeat. It seemed airtight. For a long time, the assumption that all complex life depends on oxygen felt less like a theory and more like a law of nature. It was simply how things worked.
But science has an inconvenient habit of pulling the rug out from under our certainties. Recent discoveries, some of them only published in early 2026, are forcing researchers to tear up that old rulebook. Life, it turns out, is far stranger and far more resilient than we ever imagined. Get ready, because some of what follows is genuinely hard to believe.
Life Was Never Supposed to Need Oxygen in the First Place
When life first arose, likely more than four billion years ago, there was no free oxygen in the atmosphere at all. Life was anaerobic, meaning that it did not need oxygen to live and grow. This is easy to forget when you look around at a world saturated with oxygen-breathing creatures, from redwood trees to house cats to us.
Early in Earth’s history, when the planet’s atmosphere lacked oxygen, life was strictly anaerobic. The first microbial metabolisms used electron acceptors such as carbon dioxide, iron(III), sulfate, or elemental sulfur. Think of these ancient microbes as the original survivors, powering themselves on chemical cocktails that would be toxic or useless to us today.
The evolution of cyanobacteria and the subsequent increasing oxygen concentration in the atmosphere during the Great Oxidation Event, sometime between 2.4 and 2.1 billion years ago, imposed a strong selective pressure on microbes to adapt to the presence of oxygen. In other words, oxygen was a late arrival. And not everyone welcomed it.
The Tiny Parasite That Shocked Scientists: An Animal Without Oxygen
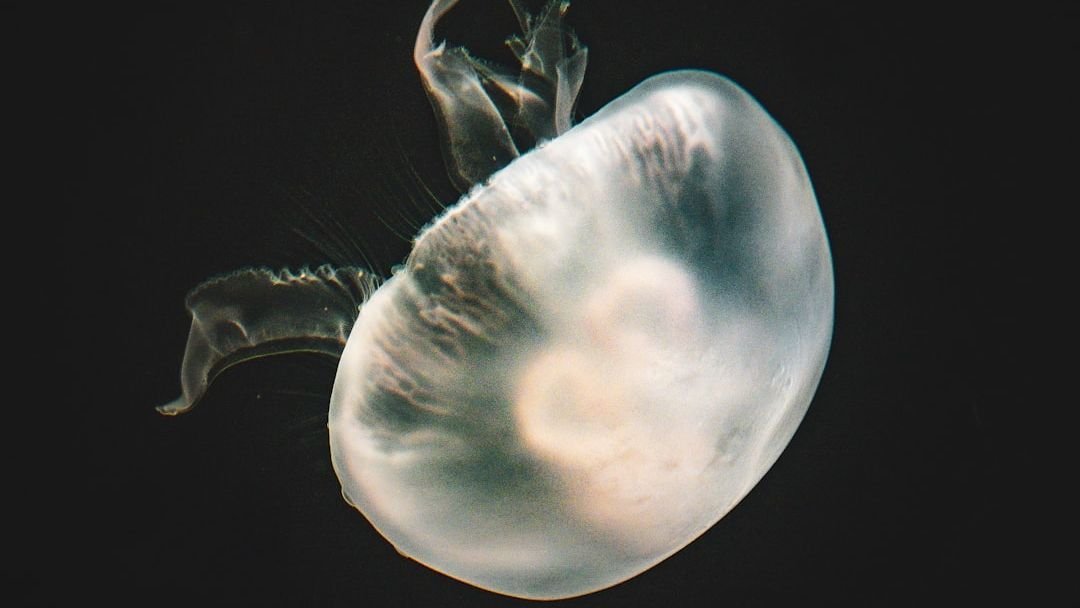
In 2020, scientists discovered a jellyfish-like parasite that doesn’t have a mitochondrial genome, the first multicellular organism ever found with such an absence. That means it doesn’t breathe; in fact, it lives its life completely free of oxygen dependency. Honestly, this finding stunned the biology community. A multicellular animal, not just a bacterium, living without oxygen altogether.
The tiny, less than ten-celled parasite Henneguya salminicola lives in salmon muscle. As it evolved, the animal, which is a myxozoan relative of jellyfish and corals, gave up breathing and consuming oxygen to produce energy. It is tucked inside its salmon host, hidden away from sunlight and air, doing just fine without either.
Aerobic respiration was thought to be ubiquitous in animals, but researchers confirmed that this is not the case. The discovery shows that evolution can go in strange directions. Aerobic respiration is a major source of energy, and yet scientists found an animal that gave up this critical pathway. It’s a bit like finding a car that runs perfectly well without an engine.
The Anaerobic World Hidden Right Inside Us
Anaerobic bacteria, which thrived in oxygen-free environments long before oxygen-dependent life forms, are vital for both human health and the ecosystem, influencing everything from gut health to disease. This isn’t some abstract deep-sea phenomenon. This is happening inside your own body, right now, while you read this.
The earth was populated with numerous organisms long before photosynthesis brought free oxygen into the world. Anaerobic bacteria have survived the ages in special, oxygen-free niches, some of them very close to us, as an essential part of the intestinal microbiome, where they are of enormous importance for the well-being of the organism. However, certain anaerobes can also trigger life-threatening diseases such as tetanus or botulism.
The AnoxyGen project led by Christian Hertweck aims to explore these bacteria’s untapped biosynthetic potentials to discover new compounds, improving medical, ecological, and biotechnological understanding and applications. The project, funded by the ERC Advanced Grant, explores the potential of anaerobic bacteria in biotechnology, medicine, and ecology. This research, launched in 2024, could lead to medical breakthroughs we haven’t even dreamed of yet.
Deep-Sea Vents: Life Fueled by Fire and Sulfur

With a rich variety of chemical energy sources and steep physical and chemical gradients, hydrothermal vent systems offer a range of habitats to support microbial life. Studies have led to an emerging view that diverse microorganisms in deep-sea hydrothermal vents live their chemolithoautotrophic, heterotrophic, or mixotrophic life with versatile metabolic strategies. Picture an environment that would kill a human instantly, and somehow find it teeming with life.
Scientists isolated species of Pyrolobus and Pyrodictium Archaea from chimney walls. These heat-loving microbes, which grow optimally at temperatures above 100°C, get their energy from hydrogen gas and produce hydrogen sulfide from sulfur compounds from the vents. These are not fringe organisms living at the edge of survival. They are thriving, dominant species in their environment.
These microbes produce energy in the absence of sunlight or oxygen through a process called anaerobic respiration, whereby inorganic compounds and ions such as protons, elemental sulfur and its derivatives, or ferric ions, are reduced to drive oxidative phosphorylation. Sulfur as a fuel source. Iron as a fuel source. It reads like science fiction, but it is very much science fact.
Microbes That Play by Their Own Rules: The Simultaneous Respirers
Recent studies reveal a more complex and fascinating reality than previously understood: some microbes can simultaneously use both aerobic and anaerobic respiration, even in fully oxic environments. Let’s be real, this one breaks the brain a little. The old model said it was either/or. Oxygen available? Use oxygen. No oxygen? Switch to anaerobic mode. Simple enough.
Some microbes can simultaneously use both aerobic and anaerobic respiration, even in fully oxic environments. This unexpected flexibility challenges classic models of microbial metabolism and sheds new light on how microbes evolved and adapted to dynamic environments. Researchers published these findings in July 2025, and the implications for our understanding of life’s adaptability are enormous.
Recent work suggests that certain microbes buck the trend of the “either/or” model of respiration. Four notable examples, including Hydrogenobacter, Synechocystis, Shewanella oneidensis, and Microbacterium deferre, demonstrate simultaneous aerobic and anaerobic respiration under conditions previously thought impossible. It’s the biological equivalent of a car running on petrol and electricity at the exact same time, not alternating, but truly simultaneously.
Life Without Mitochondria: The Loricifera Discovery
In 2010, three species of anaerobic Loricifera were discovered in the hypersaline anoxic L’Atalante basin at the bottom of the Mediterranean Sea. They lack mitochondria, which contain the oxidative phosphorylation pathway, which in all other animals combines oxygen with glucose to produce metabolic energy; thus, they consume no oxygen. Instead, these loricifera derive their energy from hydrogen, using hydrogenosomes. Hydrogen as the fuel of life. It sounds more like a rocket engine than a biology textbook.
Here’s the thing: these animals exist in a layer of brine at the bottom of the Mediterranean where there is essentially zero dissolved oxygen. The water down there is almost as hostile as it gets on this planet. Yet these creatures not only survive but apparently complete their full life cycles in complete anoxia. That challenges the very foundation of what we thought animals require to exist.
Some single-celled organisms have evolved mitochondria-related organelles for anaerobic metabolism; but the possibility of exclusively anaerobic multicellular organisms had been the subject of some scientific debate. The debate, it seems, has now been largely settled in favor of the extraordinary.
A Symbiotic Solution: Borrowing Someone Else’s Breathing Apparatus
Recently, researchers uncovered a truly remarkable example of anaerobic energy production in the deep, anoxic waters of a Swiss lake. They discovered a tiny, single-celled organism, a ciliate, living in a permanently oxygen-free zone. What’s extraordinary is that this ciliate hosts a bacterial endosymbiont, a tiny partner living inside it, that’s essentially doing the heavy lifting for energy production.
This isn’t just about survival; it’s a sophisticated partnership. The endosymbiont respires using nitrate, and in doing so, generates ATP, the universal energy currency, which it then directly provides to its ciliate host. It’s a direct transfer of energy, not just nutrients. Think of it like hiring a specialist to do all your breathing for you, then living off the paycheck they hand over.
This discovery opens up a whole new perspective, suggesting that eukaryotes with reduced mitochondria might not be limited to fermentation. They could potentially acquire specialized energy-providing endosymbionts, like this nitrate-breathing bacterium, to supplement or even replace their own mitochondrial functions. Evolution, yet again, finds a workaround we never saw coming.
The Asgard Archaea: Our Ancient Oxygen-Free Ancestors Reexamined

The most widely accepted scientific explanation for the arrival of all complex life on Earth has had an unsolved mystery at its heart. According to the theory, all plants, animals and fungi, known collectively as eukaryotes, are thought to have evolved after two very different types of microbes came together. The problem was in figuring out how the two were in such close proximity in the first place, given that one of the microbes requires oxygen for survival and the other was known to live in spaces without oxygen. Now scientists from The University of Texas at Austin, publishing in the journal Nature, appear to have solved the mystery.
Researchers used massive DNA sequencing of marine sediments to obtain 404 Asgardarchaeota metagenome-assembled genomes, including 136 new Heimdallarchaeia and several novel lineages. Analyses of their global distribution revealed they are widespread in marine environments, and many are enriched in variably oxygenated coastal sediments. Detailed metabolic reconstructions and structural predictions suggest that Heimdallarchaeia form metabolic guilds that are distinct from other Asgardarchaeota. These archaea encode hallmark proteins of an aerobic lifestyle, including electron transport chain complex IV, haem biosynthesis, and reactive oxygen species detoxification.
More than 1.7 billion years ago, oxygen levels in the atmosphere were extremely low. Then oxygen concentrations rose sharply during what scientists call the Great Oxidation Event, eventually approaching levels similar to those today. Within a few hundred thousand years of this dramatic increase, the earliest known microfossils of eukaryotes appear in the fossil record. This close timing suggests that oxygen may have played a crucial role in the emergence of complex life.
Alternative Biochemistries: Life as We Don’t Know It

How different life could be beyond Earth is still an open scientific question, and it is possible that the biochemistry of life beyond Earth could be constructed of near-identical building blocks to terran life. Nevertheless, it is reasonable to assume that distinct origins and evolutions of life in vastly different environments will yield life-forms with correspondingly different biochemistries. NASA researchers included this reasoning in their 2024 Astrobiology publication, and it is becoming increasingly mainstream thinking.
Extremophiles on Earth, organisms that thrive in extreme conditions, offer insights into how life might adapt to different environmental conditions. For instance, organisms living in hydrothermal vents have adapted to high temperatures and pressure, utilizing chemicals like hydrogen sulfide, which are toxic to most terrestrial life. These adaptations provide a model for how life might exist in non-Earth-like environments.
The biological use of sulfur as an electron acceptor is widespread and can be traced back 3.5 billion years on Earth, thus predating the use of molecular oxygen. Sulfur-reducing bacteria can utilize elemental sulfur instead of oxygen, reducing sulfur to hydrogen sulfide. That’s a metabolism older than anything we can truly comprehend, running quietly and efficiently for billions of years before oxygen even existed on this planet.
What This All Means for the Search for Life in the Universe

This discovery doesn’t just change our understanding of how life can work here on Earth; it could also have implications for the search for extraterrestrial life. That’s not a small claim. It potentially rewrites the criteria we use when scanning distant planets for signs of biology.
Current methods for detecting life are heavily biased towards Earth-like biochemistry. Instruments like spectrometers, which analyze atmospheric compositions, are calibrated for gases like oxygen and methane, which are significant for Earth-based life. This Earth-centric approach might overlook signs of life that operate on a different biochemical framework. In other words, we might have been searching for the wrong fingerprints.
The possibility of alternative biochemistries necessitates a broader definition of life, one that accommodates different physical and chemical principles. This expansion is not merely academic; it has practical implications for how we search for and recognize signs of life beyond Earth. The universe is likely far more creative than our instruments have been giving it credit for. If life here on Earth can thrive without oxygen, in boiling sulfur vents, in anoxic Mediterranean basins, and inside salmon muscle without a single mitochondrion, then what could be happening on the moons of Jupiter or in the methane lakes of Titan? What would you have guessed?